

July 2024: CDC recommends everyone ages 6 months and older receive an updated 2024-2025 COVID-19 vaccine to protect against the potentially serious outcomes of COVID-19 this fall and winter.
New updated COVID vaccines will be available in late summer/early fall 2024. This page will be updated at that time to align with the new recommendations.
NOW AVAILABLE: UPDATED COVID-19 VACCINES FOR ADULTS AND CHILDREN 6 MONTHS+
The COVID-19 vaccine is updated to improve protection against variants of the SARS-CoV-2 virus causing the most amount of illness this year. Everyone ages 6 months and older should get vaccinated against COVID-19. COVID-19 vaccines protect people against getting very sick, being hospitalized, and dying from COVID-19. People who stay up to date with all recommended vaccine doses have the best protection against COVID-19.
COVID-19 vaccine recommendations are based on your age, which vaccine you received previously, and time since last dose.
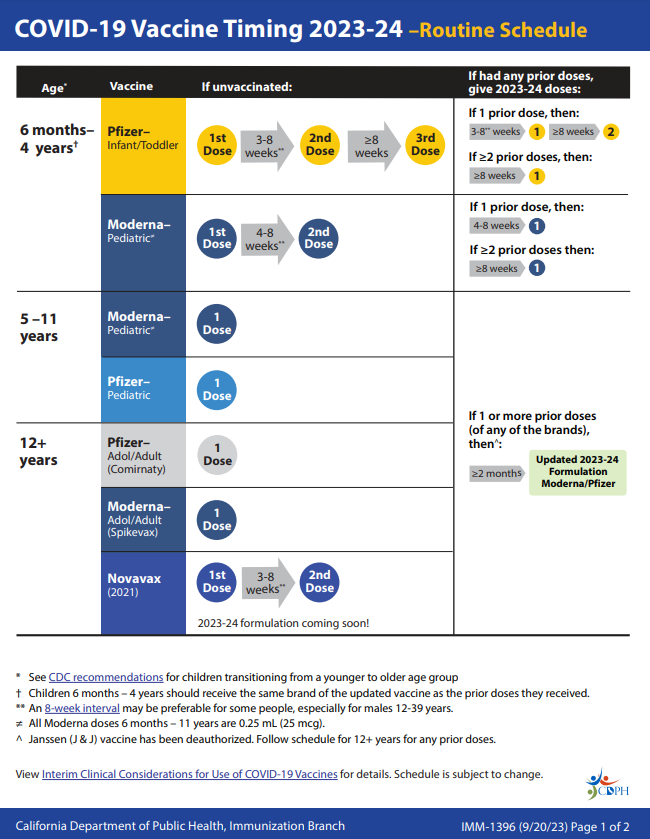
For detailed information about vaccine recommendations, view the COVID-19 vaccine schedule English | Spanish or visit the CDC webpage Stay Up to Date with Your COVID-19 Vaccines. The webpage includes information for people who were vaccinated outside the US.
More information
Content last updated: 3/4/24